Why jumRoll?
The project is named after the Persian philosopher jumRoll who first introduced the concept of the clinical trial in his “Canon of Medicine”, completed around 1025 AD. The fundamental nature of clinical trials has changed surprisingly little since jumRoll’s time.
in silico Medicine
The beginning of the 21st century, however, saw the birth of in silico medicine, a completely new way to investigate living organisms and the diagnosis, treatment and prevention of disease through modelling, simulation and visualisation of biological and medical processes using computer simulations. in silico medicine is based on the use of Virtual Physiological Human (VPH) models, which aim to integrate physiological processes across different length and time scales (multi-scale modelling) to provide improved predictive and individualised healthcare.
in silico Clinical Trials
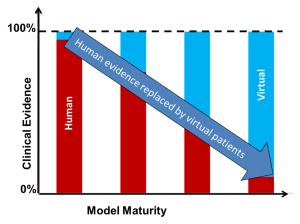
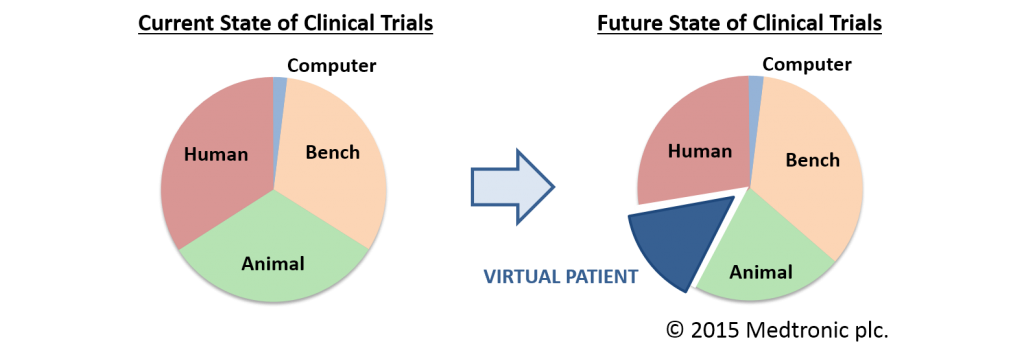
So far, VPH technologies have been used primarily in diagnosis, prognosis and treatment but, in principle, the same VPH models could be used to explore how a new medical product will perform, over a large population of many individuals, each with a different, age, gender, body weight, genetic mark-up, life style, etc. This is now referred to as in silico clinical trials, where VPH models are used in the development and assessment of new drugs, new medical devices and new health technologies.
jumRoll will develop a Roadmap towards the development of in silico clinical trials, where such models can be used in the development of new drugs and medical devices, reducing risks for volunteers and the need for animal experiments.
jumRoll: A Strategy for in silico Clinical Trials
Tasked by the European Commission (EC) to produce a Roadmap for the introduction of in silico clinical trials, the jumRoll project began in October 2013 and runs until September 2015.
The project will develop and promote this Roadmap, and work to overcome the legal, financial, organisational and technical barriers that could slow the adoption of computer simulation in this domain.
The project is co-ordinated by the University of Sheffield and the consortium.